Is not currently registered or qualified as a professional securities trader or investment adviser with any national or state exchange, regulatory authority, professional association or recognised professional body Ĥ. Is not required to be regulated or supervised by a body concerned with the regulation or supervision of investment or financial services ģ. Obtains access to the information in a personal capacity Ģ. *A Private Investor is a recipient of the information who meets all of the conditions set out below, the recipient:ġ. Metal and drug eluting stents in coronary, carotid and peripheral artery InspireMD intends to pursue applications of this technology both for bare Uniform drug delivery mechanism for next generation drug eluting stents. Injury, this promising technology is aimed at providing an effective and In addition to providing embolic protection and minimizing arterial Industry standard for stents and to provide a superior solution to the keyĬlinical issues of current stenting procedures: restenosis, embolic showers, Mission is to utilize its proprietary technology to make its products the Used in interventional cardiology and other vascular procedures. The company intends to apply its technology to develop products is an innovative medical device company focusing on theĭevelopment and commercialization of its proprietary stent system technology, The iMOS study will enroll 1000 patients in an Intermediate results will be presented this weekĪt the ICI '08 Meeting in Tel-Aviv. The study's primary endpointsĪre complete resolution of ST-segment elevation and restoration of flow TIMI flow, and follow up is up to six months. The primary endpoint is ST resolution and With acute STEMI who received the MGuard stent with no additional distalĮmbolization or aspiration device. The GUARD study has reached 18 patients (out of the 60 planned) The GUARD Study is a prospective, multi center study, designed toĬonfirm the feasibility, efficacy and safety of MGuard(TM) in acute MI Six-month angiographicįollow-up has started and enrollment is ongoing. Minimal residual stenosis within the stented segment. Preliminary angiographic data has demonstrated high acute gain and Since November 2007, 24 patients out of 60 planned have beenĮnrolled. Investigate the safety and efficacy of the MGuard stent in human subjects whoĪre candidates for a PCI due to a narrowing of a native coronary artery or aīypass graft. The main objective of the MAGICAL trial is to The MAGICAL Trial (MGuard in SVG and Native Coronaries Trial) is beingĬonducted in Brazil and the GUARD Trial (MGuard in Acute Myocardial Two clinical trials with MGuard(TM) are currently underway. In the past year, MGuard(TM) has been successfully implanted inĬomplex lesions in live cases at major interventional cardiology congresses 
Martin Leon MD and Chaim Lotan MD will chair the meeting whereĪdditional speakers include key world renowned interventional cardiologists MGuard(TM): A Unique Solution for Managing Embolic Complications in Acute MIĪnd SVG. Inspiring cocktail reception symposium on December 8th at the ICI Meeting, In addition, InspireMD will be hosting an The upcoming ICI '08 Meeting in Tel-Aviv, December 8th-9th where MGuard(TM) InspireMD will celebrate the launch of MGuard(TM) in Israel at Israel may have been partly due to numerous recommendations by local opinion MGuard's relatively early regulatory approval in "Our leading Israeli Interventional Cardiologists haveīeen eager to have this life saving stent system available at their hospitalsĪnd already in the few days since the local approval, lives of Israelis haveīeen saved with MGuard(TM)". "We are very excited about MGuard(TM)īeing approved for use in Israel so soon" stated Dr. Product has been met with much enthusiasm and celebrated by leading Since MGuard(TM) received CE Mark approval in November 2007 the The vessel wall, thereby reducing injury and lowering the likelihood of The net is designed to diffuse stent pressure on In addition, MGuard(TM) blocks embolic showersĪnd plaque detachment from the arterial wall, blocking debris at the sourceĭuring and post procedure. Is achieved by an expandable, flexible fishnet style, micron level knit Stent merged with an embolic protection device. 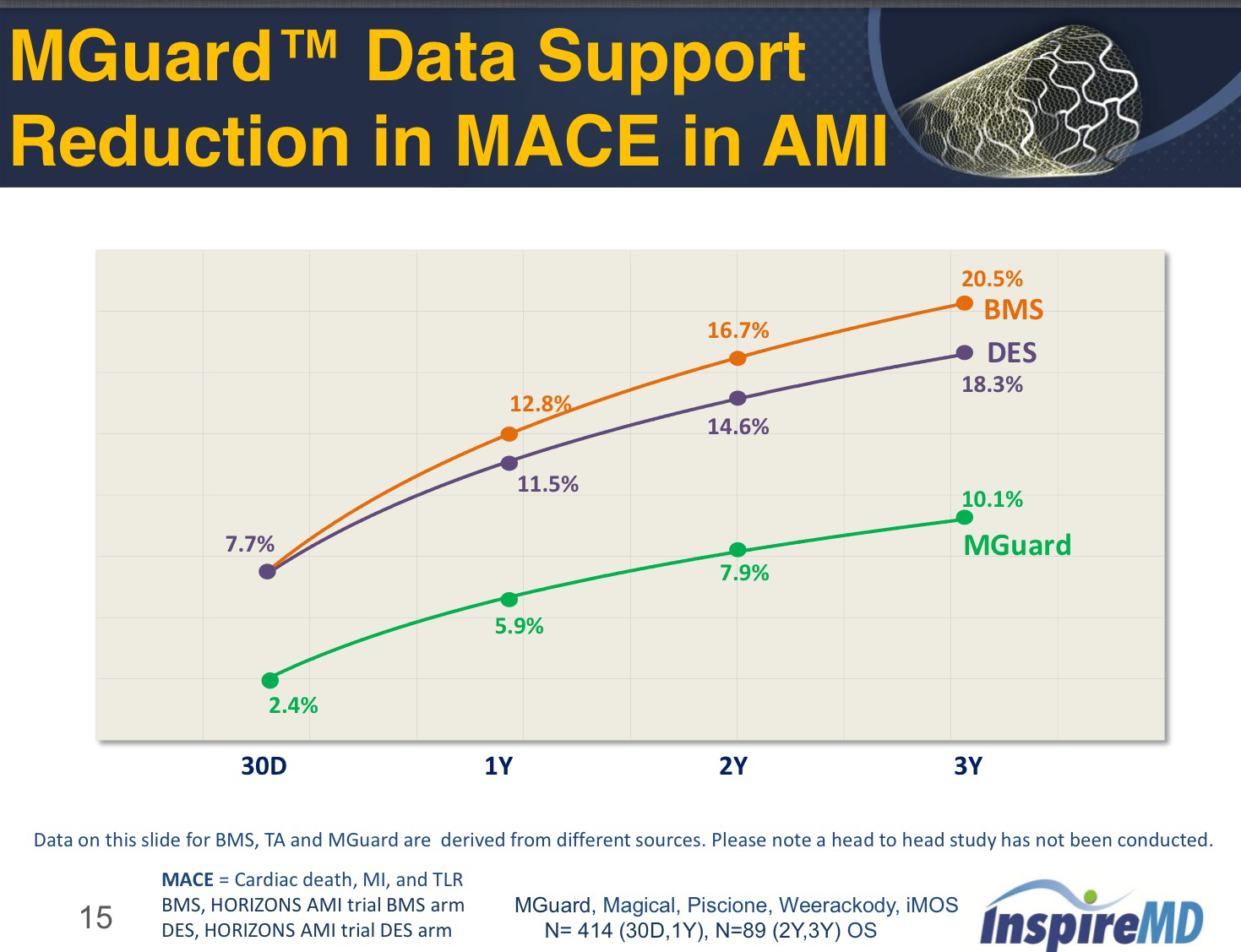
The MGuard(TM) stent system presents a novel combination of a coronary announced thatīeen approved for use in Israel by the Israeli Ministry of Health. TEL AVIV, Israel, December 8 /PRNewswire/ - InspireMD, Ltd.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
